Acids & Bases
Acids
An acid is a liquid that has a lot of hydrogen ions.
The pH range for acidic substances is 0-7
0 is the strongest
e.g citric acid, hydrochloric acid (in the stomach), lactic acid (produced on muscles when doing exercise), car batteries, fertilizer, soft drinks
Bases
A base is a liquid that has a lot of hydroxide ions.
Another word to describe bases is alkaline.
The pH range for basic substances is 7-14
14 is the strongest
Slippery
e.g cleaning products, soap, mucus (alkali), sodium hydroxide, magnesium hydroxide
The pH Scale
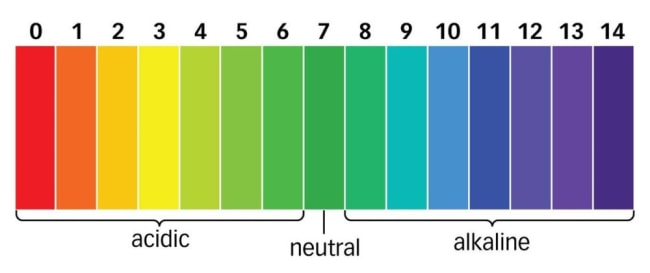
7 is neutral
The pH scale is a scale that measures how acidic or basic a liquid is.
Important Facts:
- Acids and bases can help neutralize each other
- Acids turn litmus paper red, bases turn it blue
- Strong bases can be slippery and slimy feeling (hence soap)
- Acids taste sour, bases taste bitter
- Proteins are made up of amino acids
- Vitamin C is also an acid called ascorbic acid
- Ammonia is a base chemical