Chemistry

Chemical Reactions
A chemical reaction occurs when atoms rearrange to form new substances.
- Reactants (reagents) are the substances you start with in a chemical reaction
- Products are the substances that are produced in a chemical reaction
The product will have different properties to the original reactant substances.
Signs that tell when a chemical reaction has occurred:
- A solid precipitate is formed (makes substance look cloudy)
- Gas produced (bubbles, foaming, fizzing noise)
- A substance disappears (dissolved, broken down)
- Colour change
- Light energy released
- Heat energy released (change in temperature)
- Odour
More on chemical reaction changes: click here
Chemical Equations
Reactants written on left
Products written on right
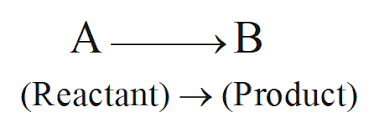
State Symbols
- (s) - solid
- (l) - liquid
- (g) - gas
- (aq) - aqueous (a solution with water as solvent)
Acids and Bases are both aqueous
Types of Reactions
Synthesis Reaction
- Two substances combine to make a new substance
- A + B --> AB
Decomposition Reaction
- Complex substance breaks down to form two separate substances
- A-B --> A + B
Combustion
- When oxygen combines with another compound to form water and carbon dioxide. Combustion reactions produce energy in the form of heat.
Single Displacement
- Also called substitute reaction
- A reaction where one compound takes a substance from another compound.
- Its equation is A + BC --> AC + B
Double Displacement
- Also called metathesis reaction
- Two compounds trading substances
- Its equation is AB + CD --> AD + CB
Photochemical Reaction
- A reaction involving protons from light
- e.g photosynthesis
- About Protons
Physical and Chemical Change
Physical Change
- Don't form new substances
- May change size or shape
- May change state of matter (melting, evaporation, freezing, solidification, sublimation)
- Are reversible (can usually be easily undone)
- Dissolving substances in a solvent to produce solutions is a physical change
- Mixtures are physical, not chemical

Chemical Change
- Involves the rearrangement of particles in the reactants to produce new substances
- Are irreversible (cannot be easily undone)
Evidence of a Chemical Reaction
- A substance disappears
- A gas is given off (bubbling, new odour produced, fizzing sound)
- A permanent colour change takes place
- A solid is precipitated
- Temperature changes (heat given off or taken in)
- Light energy produced

The Lime-water Test
The most effective way to test for CO2 is to bubble the gas through "limewater", a diluted solution of calcium hydroxide (slaked lime). When you bubble carbon dioxide through the solution, it forms a solid precipitate of calcium carbonate - chalk or limestone. Calcium carbonate is insoluble in water.
Things to Remember
Know the names and chemical formulae for the following:
(Acids)
o HCl - hydrochloric acid
o H 2 SO 4 - sulfuric acid
o HNO 3 - nitric acid
(Bases)
o NaOH - sodium hydroxide
o KOH - potassium hydroxide
o Ca(OH) 2 - calcium hydroxide