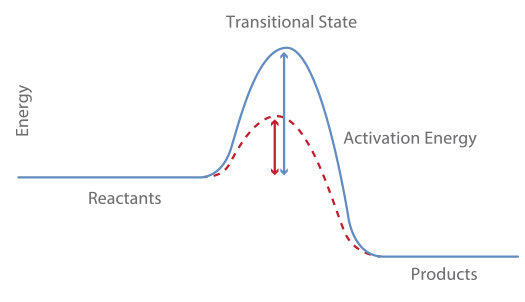
Rates of Reaction
Collision Theory - The collision theory states that for a chemical reaction to occur, the reacting particles must collide with one another.
Three things must occur in order for a chemical reaction to take place:
- Reacting particles must collide with one another
- Particles must collide with sufficient energy
- Particles must collide in the correct orientation
There are 4 ways to alter the rate of reaction:
- Temperature
- Concentration
- Particle Size
- Catalyst
Directly Proportional: When one value increases at the same rate that the other increases
Inversely Proportional: When one value decreases at the same rate that the other increases
Units to Label Data:
Molarity (M) = Used to measure concentration
Grams (g) = How much mass (e.g has been lost)
Temperature
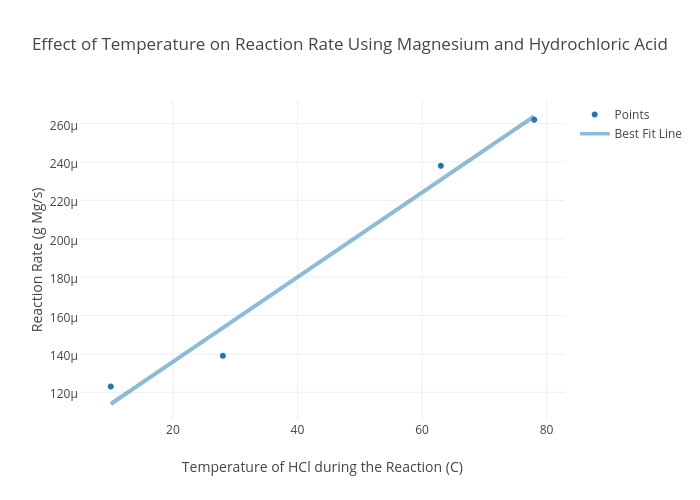
As the temperature increases, the particles gain kinetic energy and move faster. More energetic particles mean more collisions will have the minimum amount of energy needed for collision (called the "activation energy") which increases the frequency of collisions and therefore increases the rate of reaction.
There is a directly proportional relationship between the temperature and rate of reaction:
Concentration
The higher the concentration, the faster the rate of reaction. This is because there are more particles available for collision and therefore more frequent collisions. This also increases the chance of successful collisions which therefore increases the reaction rate.
Concentration is the measure of how many solute particles are in a solution. The higher the concentration means the more solute particles there are and therefore more products are formed at the end of the reaction. Concentration is usually measured in Molarity (M).
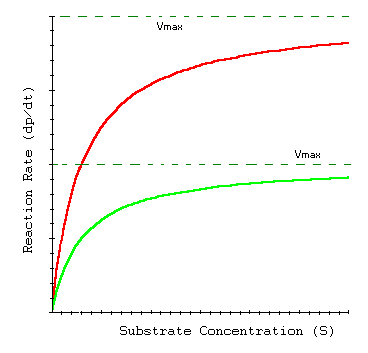
This graph shows that the high concentration reaction speeds up a lot at the start and then plateaus out earlier than the lower concentration reaction because most of the particles had already been reacted with at the start of the reaction and therefore has fewer particles to collide with later on, giving it a lower chance of colliding.
The red line represents the higher concentrated solution and the green line represents the lower concentrated solution:
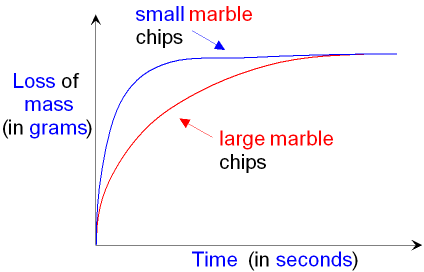
Particle Size
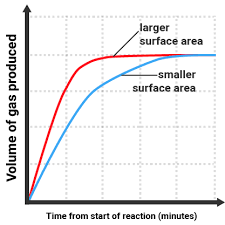
The smaller the particle size, the faster the rate of reaction. This is because there is a larger surface area and therefore creates more frequent collisions (more collisions per unit of time) so the chances of successful collisions increase.
Only the outer particles are exposed and must be reacted with in order to expose the inner particles. Smaller particle sizes have a larger surface area and have fewer particle layers, and therefore have more particles available for collision which creates more frequent collisions, increasing the rate of reaction.
Examples of graphs:
Catalyst
A catalyst can be used to alter the rate of reaction without being consumed/used up in the reaction (i.e can be reused). A catalyst can speed up the rate of a reaction by lowering the amount of activation energy needed in a reaction (called the "activation energy") which means more collisions will have sufficient energy to be successful.
The red line represents a catalysed reaction and the blue line represents an uncatalysed reaction: